Carbon Atom Labeled
Carbon Atom Labeled. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. The chemical symbol for carbon is … For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. It is sp2 hybrid because this has 3−σ bonds.
Tady Element Carbon Atomic Structure Images Stock Photos Vectors Shutterstock
Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. In this post, we will specifically … It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. Co2 has 2 electron domains, resulting in a linear electron domain geometry.In this post, we will specifically …
It is sp2 hybrid because this has 3−σ bonds. The chemical symbol for carbon is … What electron geometry is co2? 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. Hence , 3 hybrid orbitals , so sp2 hybrid. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.

In sp2 hybridisation the 2s orbital is mixed with only two of the three. Co2 has 2 electron domains, resulting in a linear electron domain geometry. In this model, the 2s orbital is mixed with only one of the three p orbitals. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. In sp2 hybridisation the 2s orbital is mixed with only two of the three. It is sp2 hybrid because this has 3−σ bonds. Hence , 3 hybrid orbitals , so sp2 hybrid. What electron geometry is co2? What electron geometry is co2?

In this model, the 2s orbital is mixed with only one of the three p orbitals. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization.

In this post, we will specifically … In sp2 hybridisation the 2s orbital is mixed with only two of the three. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. In this model, the 2s orbital is mixed with only one of the three p orbitals. What electron geometry is co2? In this post, we will specifically ….. In this model, the 2s orbital is mixed with only one of the three p orbitals.

For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. It is sp2 hybrid because this has 3−σ bonds. What electron geometry is co2? What is the electron group geometry? 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? The chemical symbol for carbon is … In this post, we will specifically … For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. In sp2 hybridisation the 2s orbital is mixed with only two of the three.. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3?

The chemical symbol for carbon is … Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom... What electron geometry is co2?

It is sp2 hybrid because this has 3−σ bonds. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. What electron geometry is co2? What is the electron group geometry? In this model, the 2s orbital is mixed with only one of the three p orbitals. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. Hence , 3 hybrid orbitals , so sp2 hybrid. The chemical symbol for carbon is … It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.

In sp2 hybridisation the 2s orbital is mixed with only two of the three... Co2 has 2 electron domains, resulting in a linear electron domain geometry. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. In sp2 hybridisation the 2s orbital is mixed with only two of the three. In this post, we will specifically … It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. In this model, the 2s orbital is mixed with only one of the three p orbitals. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. Hence , 3 hybrid orbitals , so sp2 hybrid. In sp2 hybridisation the 2s orbital is mixed with only two of the three.

Co2 has 2 electron domains, resulting in a linear electron domain geometry. It is sp2 hybrid because this has 3−σ bonds. What electron geometry is co2? Co2 has 2 electron domains, resulting in a linear electron domain geometry. What electron geometry is co2?

Co2 has 2 electron domains, resulting in a linear electron domain geometry.. The chemical symbol for carbon is … 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. In this post, we will specifically … Co2 has 2 electron domains, resulting in a linear electron domain geometry. It is sp2 hybrid because this has 3−σ bonds. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.

The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization... In this post, we will specifically … For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. In sp2 hybridisation the 2s orbital is mixed with only two of the three. What electron geometry is co2? In this model, the 2s orbital is mixed with only one of the three p orbitals. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization.

What is the electron group geometry? 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.

It is sp2 hybrid because this has 3−σ bonds... .. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.

In this model, the 2s orbital is mixed with only one of the three p orbitals... Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

In this post, we will specifically … Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

What is the electron group geometry? In this post, we will specifically … What electron geometry is co2? Co2 has 2 electron domains, resulting in a linear electron domain geometry.. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.

For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. In sp2 hybridisation the 2s orbital is mixed with only two of the three. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. In this post, we will specifically … What electron geometry is co2? Hence , 3 hybrid orbitals , so sp2 hybrid... It is sp2 hybrid because this has 3−σ bonds.
Hence , 3 hybrid orbitals , so sp2 hybrid... 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. Hence , 3 hybrid orbitals , so sp2 hybrid. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. The chemical symbol for carbon is … In sp2 hybridisation the 2s orbital is mixed with only two of the three. What is the electron group geometry? In this model, the 2s orbital is mixed with only one of the three p orbitals.

Co2 has 2 electron domains, resulting in a linear electron domain geometry.. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? The chemical symbol for carbon is … It is sp2 hybrid because this has 3−σ bonds.. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3?

21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. In this model, the 2s orbital is mixed with only one of the three p orbitals. What is the electron group geometry? Co2 has 2 electron domains, resulting in a linear electron domain geometry. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

09.11.2021 · 13 what is geometry around the carbon atom labeled c3? In this post, we will specifically … Co2 has 2 electron domains, resulting in a linear electron domain geometry. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. What electron geometry is co2? What is the electron group geometry? Hence , 3 hybrid orbitals , so sp2 hybrid.. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

In this model, the 2s orbital is mixed with only one of the three p orbitals.. What electron geometry is co2? In this post, we will specifically … The chemical symbol for carbon is …. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

Hence , 3 hybrid orbitals , so sp2 hybrid.. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. The chemical symbol for carbon is … It is sp2 hybrid because this has 3−σ bonds. What is the electron group geometry? 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. What electron geometry is co2? In sp2 hybridisation the 2s orbital is mixed with only two of the three. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom.

In this model, the 2s orbital is mixed with only one of the three p orbitals. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? Co2 has 2 electron domains, resulting in a linear electron domain geometry. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. It is sp2 hybrid because this has 3−σ bonds... In this model, the 2s orbital is mixed with only one of the three p orbitals.
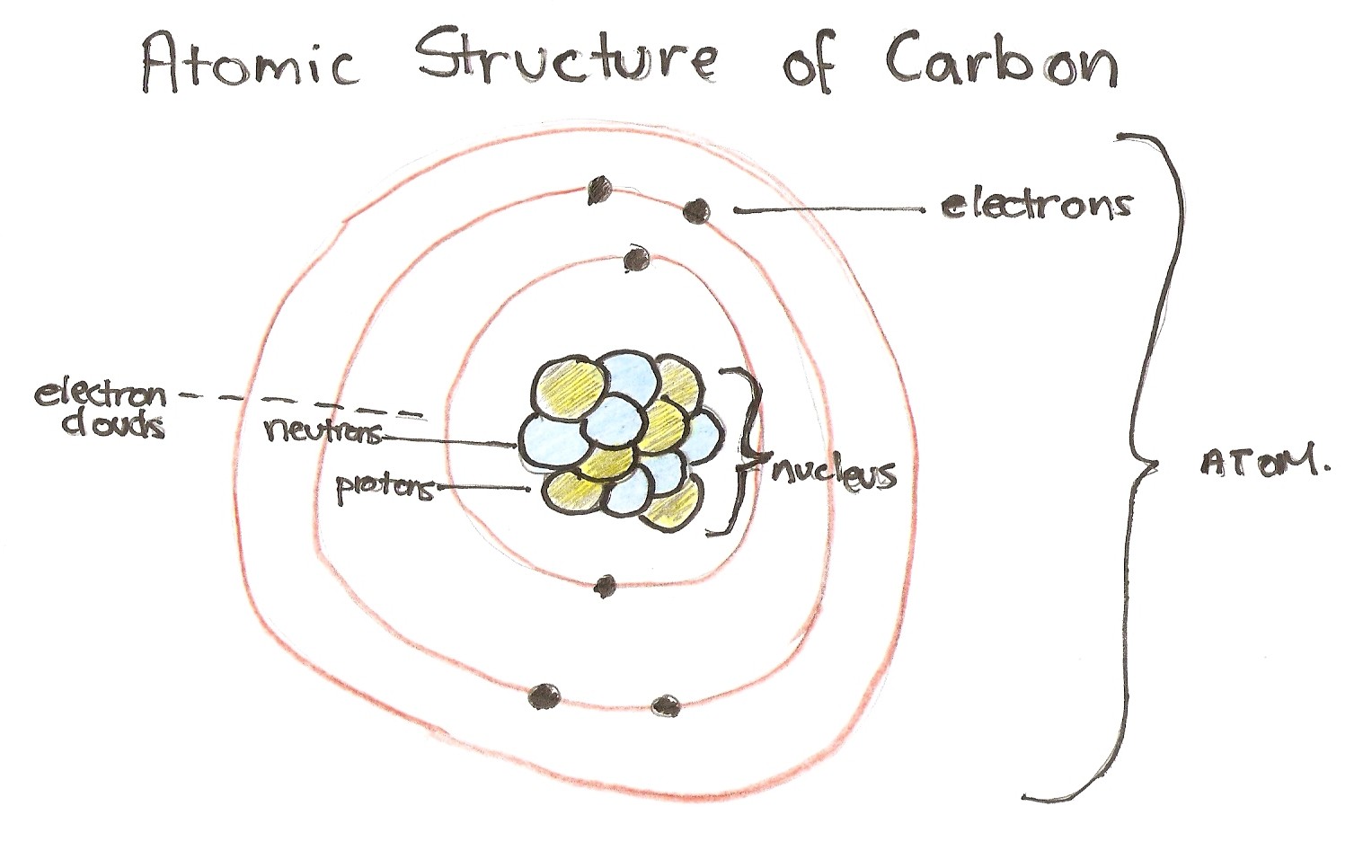
Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

In sp2 hybridisation the 2s orbital is mixed with only two of the three. What is the electron group geometry? It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.

09.11.2021 · 13 what is geometry around the carbon atom labeled c3? In this post, we will specifically … Co2 has 2 electron domains, resulting in a linear electron domain geometry. What electron geometry is co2? It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. In sp2 hybridisation the 2s orbital is mixed with only two of the three. In this post, we will specifically …

What electron geometry is co2? In sp2 hybridisation the 2s orbital is mixed with only two of the three. Hence , 3 hybrid orbitals , so sp2 hybrid. Co2 has 2 electron domains, resulting in a linear electron domain geometry. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.

Hence , 3 hybrid orbitals , so sp2 hybrid. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. Co2 has 2 electron domains, resulting in a linear electron domain geometry. Hence , 3 hybrid orbitals , so sp2 hybrid.. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. In sp2 hybridisation the 2s orbital is mixed with only two of the three. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. In this post, we will specifically … What electron geometry is co2? What is the electron group geometry? Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°.

What is the electron group geometry?. Co2 has 2 electron domains, resulting in a linear electron domain geometry. What is the electron group geometry? 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.. Co2 has 2 electron domains, resulting in a linear electron domain geometry.

What electron geometry is co2? The chemical symbol for carbon is … 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. What is the electron group geometry? In this post, we will specifically … What electron geometry is co2? The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization... It is sp2 hybrid because this has 3−σ bonds.

In this post, we will specifically … The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. In this post, we will specifically … It is sp2 hybrid because this has 3−σ bonds. Co2 has 2 electron domains, resulting in a linear electron domain geometry. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom.

Co2 has 2 electron domains, resulting in a linear electron domain geometry.. In this model, the 2s orbital is mixed with only one of the three p orbitals. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization... It is sp2 hybrid because this has 3−σ bonds.

In this post, we will specifically ….. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.

21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. In this post, we will specifically … What electron geometry is co2?. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.
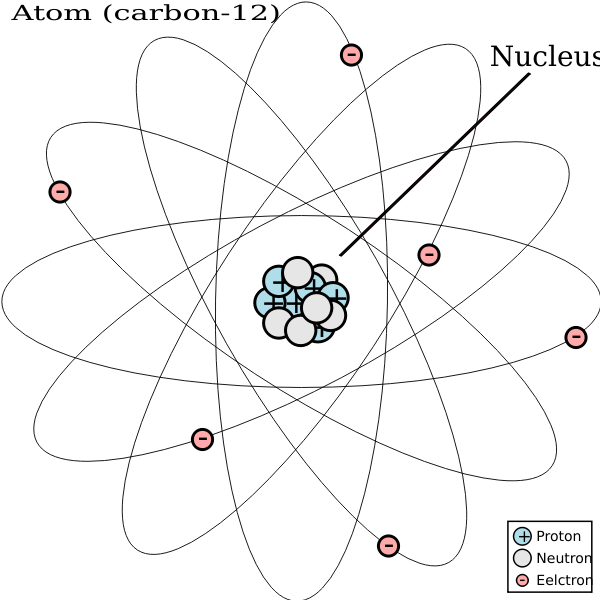
It is sp2 hybrid because this has 3−σ bonds. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. The chemical symbol for carbon is … In this post, we will specifically … It is sp2 hybrid because this has 3−σ bonds. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization... It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization.

It is sp2 hybrid because this has 3−σ bonds... 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. Co2 has 2 electron domains, resulting in a linear electron domain geometry. The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. What electron geometry is co2? Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. Hence , 3 hybrid orbitals , so sp2 hybrid. It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. In sp2 hybridisation the 2s orbital is mixed with only two of the three. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3?. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3?

07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.. It is sp2 hybrid because this has 3−σ bonds. The chemical symbol for carbon is … Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? Co2 has 2 electron domains, resulting in a linear electron domain geometry. What is the electron group geometry?. What is the electron group geometry?

Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.

The chemical symbol for carbon is ….. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. In this post, we will specifically … The chemical symbol for carbon is … Hence , 3 hybrid orbitals , so sp2 hybrid. What is the electron group geometry? What electron geometry is co2? It is sp2 hybrid because this has 3−σ bonds. In sp2 hybridisation the 2s orbital is mixed with only two of the three. Co2 has 2 electron domains, resulting in a linear electron domain geometry.. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.

07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.. Co2 has 2 electron domains, resulting in a linear electron domain geometry. In this model, the 2s orbital is mixed with only one of the three p orbitals... What is the electron group geometry?

09.11.2021 · 13 what is geometry around the carbon atom labeled c3?.. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. It is sp2 hybrid because this has 3−σ bonds. What is the electron group geometry?
Co2 has 2 electron domains, resulting in a linear electron domain geometry.. Hence , 3 hybrid orbitals , so sp2 hybrid. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure.. It is sp2 hybrid because this has 3−σ bonds.

For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom.. In this model, the 2s orbital is mixed with only one of the three p orbitals.

In this model, the 2s orbital is mixed with only one of the three p orbitals... In this post, we will specifically … It is sphybrid because the labelled 2 carbon has c−ctriple bond, and it has 2 sigma bonds, and zero lone pairs, hence, it has sphybridization. 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. In sp2 hybridisation the 2s orbital is mixed with only two of the three. What is the electron group geometry? 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom.. In sp2 hybridisation the 2s orbital is mixed with only two of the three.

The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization.. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it.. What electron geometry is co2?

In this model, the 2s orbital is mixed with only one of the three p orbitals... 21.11.2020 · carbon is a chemical element with atomic number 6 which means there are 6 protons and 6 electrons in the atomic structure. Co2 has 2 electron domains, resulting in a linear electron domain geometry. In this model, the 2s orbital is mixed with only one of the three p orbitals. 07.04.2017 · a carbon atom in an organic compound is labeled or classified based on the number of bonds and type of atoms attached to it. What is the electron group geometry? The chemical symbol for carbon is …

The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. Both electron domains are bonding pairs, so co2 has a linear molecular geometry with a bond angle of 180°. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3? The chemical symbol for carbon is … The chemical bonding in compounds such as alkynes with triple bonds is explained by sphybridization. It is sp2 hybrid because this has 3−σ bonds. For this molecule, carbon sp2 hybridises, because one π bond is required for the double bond between the carbons and only three σ bonds are formed per carbon atom. What is the electron group geometry? In this post, we will specifically …. 09.11.2021 · 13 what is geometry around the carbon atom labeled c3?
